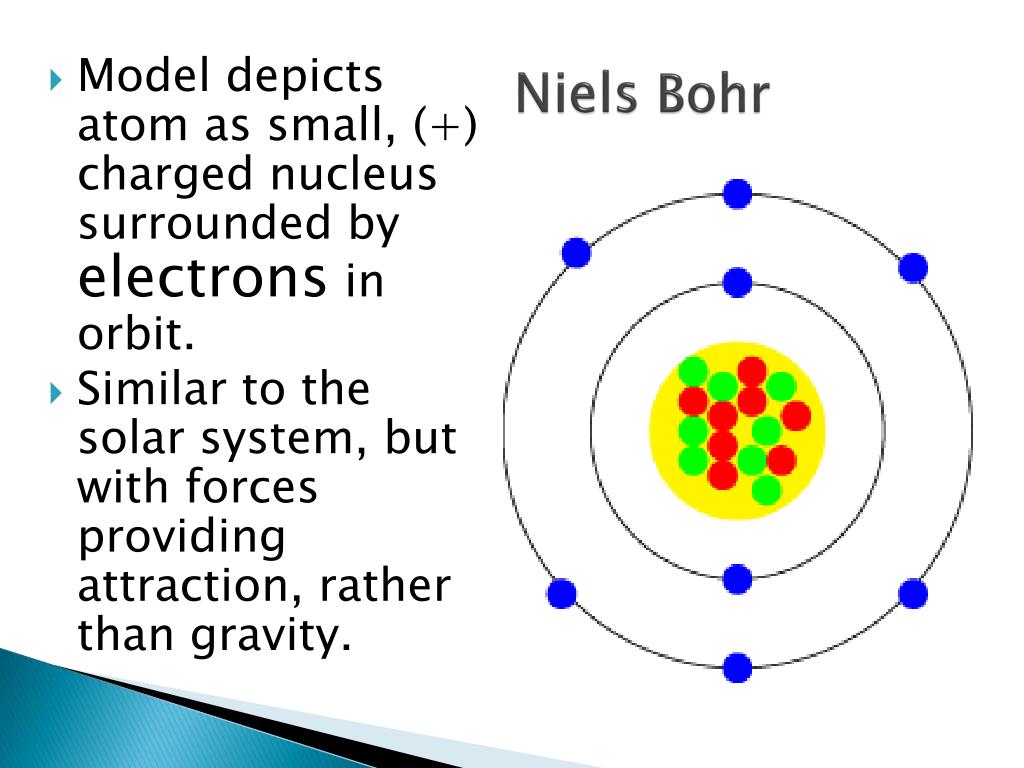
Our new and comprehensive learning solutions are designed to bring science to life by giving you all the tools you need to incorporate more (and effective) hands-on learning in your classroom or lab. We’re introducing a whole new way to teach experiential science! We’ve taken a hard look at science study from all perspectives and, thanks to the feedback of thousands of teachers nationwide, we’ve created a way to make it easier to provide personalized instruction while delivering the kind of lab experiences your students truly need. Our 3-year option extends access to digital content to three years, with a 30% discount on refill supply kits years 2 and 3. /bohr-atomic-model-141482466-5c39863dc9e77c000129ff8d.jpg)
Our 1-year option provides access to all digital content for one year plus lab supplies for a single class of 30 students. With a few simple and safe materials, your students are sure to grasp this important concept!Īssociated Phenomena: What causes the colors in a fireworks display? Quantum mechanics deals only with the probability of finding a particle within a given region of space at any given time. Heisenberg’s uncertainty principle states that there is a fundamental limitation as to just how precisely both the position and the momentum of a particle can be known at any given time. Outcomes: Students explore the difficulty of finding an electron around the nucleus and come to understand that we can only determine the probability of its location. 360Science™: Evaluate the Bohr Model of the AtomĬoncepts: Quantum mechanics, electron energy levels, Heisenberg’s uncertainty principle, orbital shapes
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
